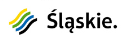The Wavelength of Corrosion
As part of the experiment “The Wavelength of Corrosion,” we investigated how contact between strings and human skin affects their degradation and sound - before and after the corrosion process. The results turned out to be intriguing.
Over the past few months, we conducted detailed research, the findings of which were published in a popular science article in the bulletin of the Silesian University of Technology.
Curious what corrosion sounds like?
Corrosion has its own sound. Can you hear it?
If corrosion could speak, would you hear its words? Or perhaps… its sound? Have you ever wondered how the sound of brand-new guitar strings differs from those that have undergone hundreds of hours of playing?
Although the changes occurring on strings are often invisible to the naked eye, their impact can be audible. Sweat, oils, and contaminants transferred from a musician’s fingers during playing significantly accelerate the corrosion process, leading to a gradual deterioration of sound quality. Is this the only effect of the degradation of such a crucial component of the instrument - one that is essential for producing sound from the wooden body?
Construction of Guitar Strings
Modern guitar strings are complex metallic structures, most commonly consisting of a core with a hexagonal or round cross‑section made of high‑carbon steel, and a winding of nickel‑plated steel wire or copper alloys such as phosphor bronze (Fig. 1).
The core is often coated with a thin layer of tin (Sn), which is intended to limit stress corrosion and improve the sliding of the winding, while the outer nickel coating gives the sound a “warmer” tone and protects the string from rapid wear. Corrosion processes are initiated through contact with human sweat. Electrochemical reactions lead to the release of metal ions into the surrounding environment, such as iron (Fe³⁺), tin (Sn²⁺), manganese (Mn²⁺), and nickel (Ni²⁺). Of particular concern for musicians’ health is the release of nickel, a strong allergen that can cause contact dermatitis, with lactic acid present in sweat further enhancing its solubility via chelation processes.
Studies conducted by the research team led by Professor Rezić at the University of Zagreb, involving measurements of selected ion concentrations in a solution of simulated artificial sweat, demonstrate that the release of nickel ions from electric guitar strings (Fig. 2) is a real problem. However, during the course of the experiment, no exceedance of the concentration generally accepted by regulations as hazardous in terms of inducing sensitization (> 0.5 μg/cm²/week) was observed.

Progressive corrosion of guitar strings drastically affects their acoustic properties, causing attenuation of higher harmonics and shortening of the instrument’s sustain. Corrosion products (in the case of iron/steel - rust) accumulating in the gaps of the winding increase internal friction and damping, which manifests as a loss of kinetic energy of the vibrating string and results in a less “bright,” duller sound lacking brilliance. To counteract these negative effects, manufacturers employ, among other solutions, polymer coatings that isolate the metal from external factors, or they use noble metals such as gold or silver.
Recent studies conducted under the supervision of Professor Cases at the Polytechnic University of Valencia also indicate the high effectiveness of external‑current cathodic protection, which makes it possible to reduce the formation of iron oxides by approximately 40%, effectively extending string lifespan and allowing their original tonal qualities to be preserved for a longer time (Fig. 3). In one of the studies led by Cases, spectrograms (color plots with horizontal lines) were recorded, illustrating how the intensities of individual sound frequencies change over time following a controlled excitation of a guitar string (the redder a point on the plot, the higher the intensity of a given frequency; the greener the point, the lower the intensity). The study also published FFT spectra (Fast Fourier Transform), defining the intensity (amplitude) of individual frequencies at a single moment in time (representing a single slice of the spectrogram).

As can be seen from the presented graphs, corrosion of an electric guitar string not only reduces the amplitude of the sound (its loudness), but also alters the FFT spectrum (changes the timbre) and shortens the sustain (the time over which the sound resonates).
Our Approach to Research on the Corrosion of Strings
On the occasion of this year’s CorrDay, we decided to contribute our own input to the global research on the corrosion of guitar strings. We know that string degradation leads to the release of harmful ions and to the “erosion” of an instrument’s sound. It was this latter aspect that particularly interested us, as quantitative information on changes in sound characteristics (e.g., Fig. 3) is available, but we were unable to find qualitative descriptions. In other words, we wanted to listen to corrosion. How do brand‑new strings sound compared to corroded ones?
Additionally, there is a noticeable focus in the scientific community on the corrosion of strings intended exclusively for electric guitars. For this reason, we decided to include an acoustic guitar in our study as well.
To conduct the experiments, we selected two sets of strings from the popular guitar brand D’Addario (Fig. 4):
- EXL 110 (electric guitar; corresponding to a string gauge range of 10–46; steel, nickel‑plated strings)
- EJ15 (acoustic guitar; corresponding to a string gauge range of 10–47; steel strings with phosphor bronze winding)

Fig. 4. Two sets of strings suspended in the laboratory for research purposes: (left) appearance of the strings, (center) strings placed in pockets filled with an artificial sweat solution, (right) close‑up of the labeled pockets. (photos: A. Kołkowska)
We immersed the strings in an artificial sweat solution 5 days a week for 30 minutes per day over the course of 3 weeks, in order to simulate the conditions under which beginner guitarists practice and develop their skills. We will not hide the fact that these conditions were rather extreme - after all, no one is capable of sweating so much onto their strings that each of them becomes covered with 100 ml of such a solution, which is precisely the volume we used. However, thanks to this immersion procedure, three weeks were sufficient to observe noticeable effects!
You are probably wondering what an artificial sweat solution actually is. It turns out that there is even a standard that strictly regulates its composition (for those interested, the standard number is EN 1811:2023). Such a solution is used in laboratories, for example when conducting experiments related to the release of metal ions that may potentially cause allergic reactions (this is of interest, for instance, to the jewelry industry). To prepare artificial sweat, the following components must be added to water in the specified proportions:
- 0.5 wt% NaCl (sodium chloride - common table salt and the main corrosive agent)
- 0.1 wt% lactic acid (minimum 88% purity; commonly and mistakenly believed to be the cause of muscle soreness, or DOMS, after exercise)
- 0.1% urea (commonly found in moisturizing creams, e.g., for foot care)
- ammonium solution used to adjust the pH of the solution to 6.5 (close to neutral)
After completing the “sweating” experiments on the strings, we proceeded to examine our “patients” in the following ways:
- changes in string weight
- determination of instantaneous corrosion resistance (determined electrochemically - more on this later)
- changes in string appearance examined using a scanning electron microscope (SEM)
- assessment of the strings’ sound
Unfortunately, changes in weight could not be determined without removing the corrosion products (mainly rust), which were necessary for examining their influence on sound. For this reason, we will not be sharing these results. This is because, in place of the corroded metal, an insoluble deposit forms that has a similar mass - at least from the perspective of the accuracy of our laboratory balances.
To conduct electrochemical tests, we had to create an entirely new measurement setup. Such studies are performed in a vessel filled with a corrosive medium (in this case, the artificial sweat solution) into which electrodes are introduced (a reference electrode and an auxiliary electrode, for those curious). Our object of study (the string) also became one of the electrodes (known as the working electrode). In order for the string to be properly tensioned (to its natural tuning), we had to use a device that allowed us to obtain a specific musical note in a repeatable manner. This gave rise to what we ultimately called a “measurement instrument” (Fig. 5).

Fig. 5. Measurement instrument used in the research: (left) the measurement instrument connected to an AUTOLAB potentiostat–galvanostat; (right) close‑up of the measurement vessel (the electrochemical cell) with electrodes. The well‑worn acoustic guitar was donated for scientific purposes by A. Olesiński. (photos: A. Kołkowska)
Studies using a potentiostat-galvanostat (Fig. 5) involve precise control of electrical parameters (current and potential) in order to determine appropriate corrosion characteristics. For this purpose, a method known as electrochemical impedance spectroscopy (EIS) can be applied. Without going into details, this technique allows the determination of the polarization resistance, whose numerical value (expressed in units of Ω·cm²) is proportional to the material’s instantaneous corrosion resistance. Such measurements were carried out on selected strings before immersion in the artificial sweat solution and after each week of this “sweating” process (Fig. 6).
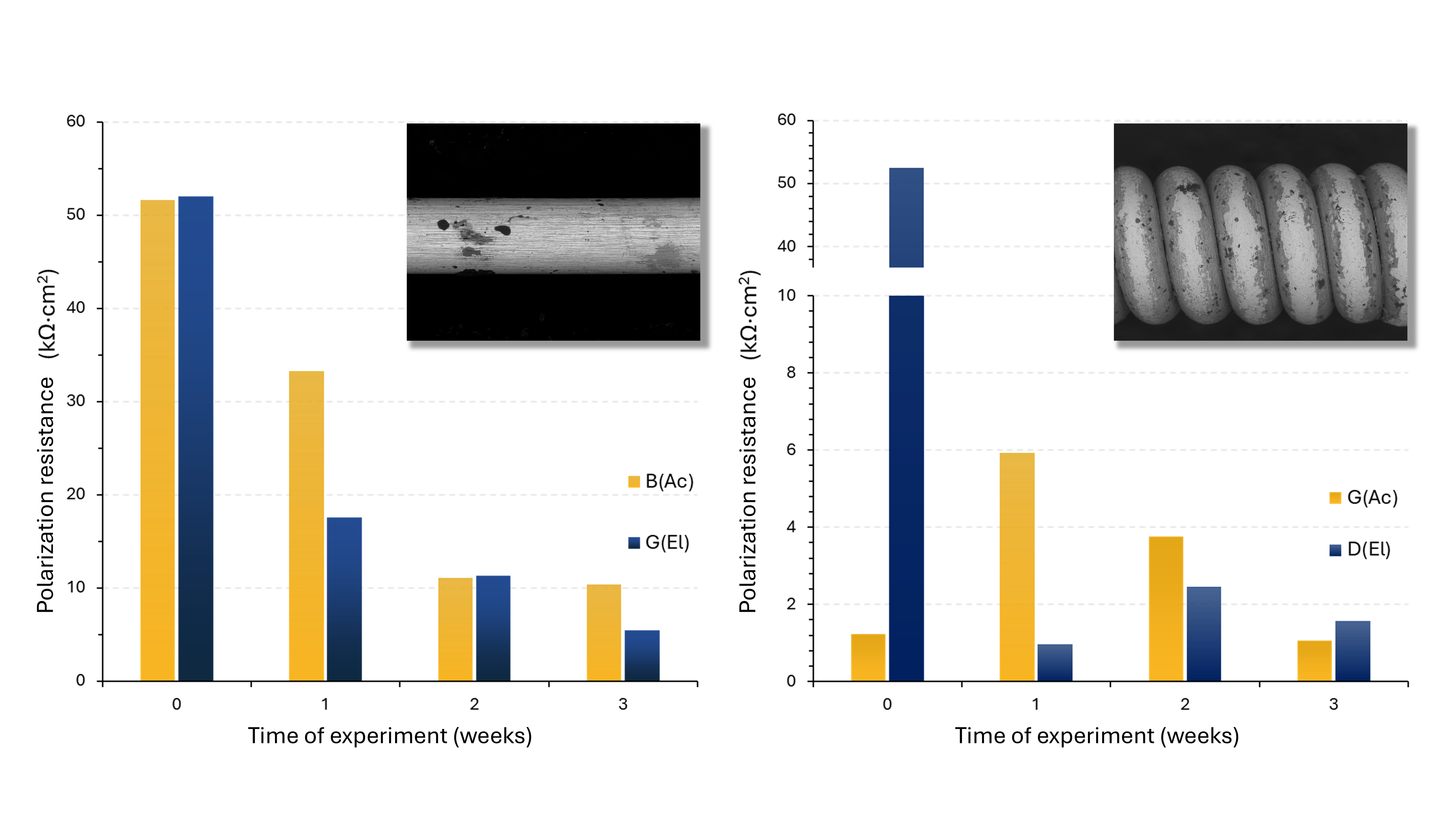
Fig. 6. Instantaneous corrosion resistance, expressed as polarization resistance, determined for the B2(Ac) string - unwound and G3(Ac) - wound on an acoustic guitar, as well as G3(El) - unwound and D(El) - wound on an electric guitar. The insets show SEM images of electric guitar strings (left: G(El), right: D(El)) after 3 weeks of corrosion.
The corrosion resistance shown in the plots (Fig. 6) is normalized by the surface area of the strings, which means that their size (thickness) does not affect the value of this parameter. We can see that both unwound strings (left plot) exhibit very similar behavior, regardless of whether they are used on an acoustic or an electric guitar. This is hardly surprising - they are made of the same material, differing only in thickness. Over time, however, the resistance decreases, and the first traces of rust appear on the string (inset with a microscopic image in the left plot of Fig. 6).
The situation is completely different for wound strings (right plot). In the case of the electric guitar string, its initial resistance value is identical to that of the unwound strings (well done, electric guitar!). However, for the wound acoustic guitar string, the polarization resistance is about 50 times lower compared to the other strings. This is caused by phosphor bronze and the phenomenon of galvanic corrosion. Now a bit of technical explanation - I'll try not to get carried away. Everything depends on the nobility of metals that are in direct contact. For example, gold is very noble, copper is moderately noble, iron is of low nobility, and sodium is extremely non‑noble (better not meet it in a dark alley...). The greater the difference in nobility between two metals, the more the more noble partner accelerates the corrosion of the less noble one. At the same time, the more noble partner corrodes more slowly in such company than it would alone in the same environment (apparently, being noble pays off!). The copper contained in the phosphor bronze winding is relatively noble, so it strongly accelerates the corrosion of the iron core. However, as can be seen, over time the buildup of corrosion products slows this effect (inset with a microscopic image in the right plot of Fig. 6). Nickel, on the other hand (although moderately more noble than iron) additionally forms a passive layer that separates it from its partner, resulting in a smaller initial acceleration. Over time, however, an equalization of the resistance of both strings becomes visible, revealing the effect of galvanic corrosion (compare with the left plot for unwound strings). Phew… end of the technicalities. In short: the winding improves sound quality and enables the production of lower frequencies without making the string excessively stiff, but it also introduces corrosion issues.
Let us now take a look at the strings after cutting them in half (yes, it can be done - Fig. 7A; especially with a sufficiently large number of students/PhD candidates). For this purpose, we use a special SEM‑EDS variant (energy‑dispersive X‑ray spectroscopy), which allows visualization of the elements present in the material (Fig. 7).

Fig. 7. (A) Cross‑sections of the G(Ac) acoustic guitar string before and after corrosion – macroscopic view. Microscopic images of the post‑corrosion cross‑sections together with elemental distribution maps for (B) the B(Ac) acoustic guitar string, (C) the D(El) electric guitar string, and (D) the G(Ac) acoustic guitar string.
Microscopic images of string cross‑sections reveal the material composition of the observed regions. Elemental distributions are shown as colorful “maps” placed next to the gray images from the scanning electron microscope (electrons do not allow colors to be visualized; the colors we see on a daily basis are made possible by light, i.e., photons). In the case of the B(Ac) acoustic guitar string, only faint traces of corrosion can be seen at the edges, especially in its upper part (Fig. 7B). The remainder of the material consists mainly of iron. Surrounding signals originate from carbon and oxygen, which make up the resin in which the strings were embedded for observation. For the wound D(El) electric guitar string (Fig. 7C), traces of corrosion are visible in limited amounts between the turns of the winding at the contact area with the core. However, they are not very numerous. What is clearly visible is that the winding is indeed nickel‑plated. The most severe damage is observed in the wound G(Ac) acoustic guitar string (Fig. 7D). In addition to differences in the chemical composition of the core (iron) and the winding (a copper–tin alloy, i.e. bronze, more precisely phosphor bronze), corrosion products (rust) rich in iron and oxygen are visible both on the outer surface and beneath the winding. It is precisely the galvanic effect that makes this material combination exhibit the most severe corrosion after a three‑week “sweating” cycle.
Degradation of Sound
Now comes the best part. We will have the opportunity to listen to how the strings sound after their aforementioned encounter with the artificial sweat solution, compared with brand‑new strings. Sound samples of each open string (i.e., played without changing pitch by pressing against the frets) were recorded on both the electric and acoustic guitars (using headphones is highly recommended).
Although we are primarily chemical engineers and guitar‑playing enthusiasts, we decided to undertake a very basic analysis of the acoustic signal obtained from recordings of the guitars in these four different configurations. Below is a summary of the FFT spectra (discussed at the beginning of this article) recorded for all the strings examined in our study (Fig. 8).

Fig. 8. FFT spectra recorded for open strings on the acoustic guitar (left) and the electric guitar (right). Signals shown in yellow can be attributed to corroded strings, while those in orange correspond to new strings. The green line represents an envelope connecting the intensity peaks of the new strings.
Based on the analysis of the frequency spectra above, it can be concluded that corrosion processes have a more pronounced effect on the acoustic properties of wound strings than on plain strings. In the case of corroded strings, a significant reduction in the amplitude of the fundamental frequency (the frequency to which the string is tuned) is observed compared to brand-new strings, indicating a deterioration in the efficiency of the string’s natural vibrations.
For unwound strings, this relationship is much weaker and in many cases almost imperceptible. This is consistent with expectations, as wound strings contain spaces that promote the accumulation of corrosion products, contaminants, and skin residues, which can lead to increased vibration damping and changes in the effective mass of the string. From the above studies, we also know that galvanic corrosion (nickel-steel and phosphor bronze-steel) contributed to faster corrosive degradation than in the case of free corrosion (unwound strings).
Additionally, it was observed that for the B2 and e1 strings on the electric guitar, the amplitude of the fundamental frequency is relatively low for both new and corroded strings. This may indicate the presence of factors not directly related to corrosion, such as suboptimal bridge adjustment or unwanted friction at the nut, which can limit the free vibration of the string.
If what you have read, seen, and heard above seems too raw, cold, or lacking character, we have something more - something that will allow you to truly feel the difference between fresh and corroded strings. The best way to do this is through an example: a musical piece that will serve as a basis for comparing the sound of instruments whose strings are affected (or unaffected) by corrosion.
In Tribute to the Masters
In connection with the upcoming (19.05.2026) concert of the global guitar powerhouse - the American band Metallica (who surely need no introduction…), we decided to choose one of their original ballad compositions: Fade to Black. You can judge for yourselves the sound of this melancholic ballad played on brand-new and corroded strings. In our opinion, the corrosion that plagues the world fits perfectly with the song’s lyrical theme.
Which version of the recording did you like more?
If that’s still not enough for you…
…and if you haven’t had the chance yet, we invite you to listen to a podcast (in Polish, sorry...) featuring the main instigators of this small corrosion‑focused project:
- mgr inż. Aleksander Olesiński
- dr inż. Maciej Sowa
Thanks to the hospitality of ed. Martin Huć, a member of the team producing the Silesian University of Technology podcast “Let’s Talk Science”, we were given a platform where we could casually discuss the origins of the experimental idea and our musical passions.
On 24.04.2026, when we held a World Corrosion Awareness Day Ceremony (WCAD or CorrDay 2026 at SUT), the gathered audience had a chance to listen the strings first-hand. What is even cooler, they had a chance to literally leave their mark on the "measurement instrument" (Fig. 5). Now, this scientifically-proven, corrosion-tested guitar will be yet another mascot of the CorrDay events to come (Fig. 9).
See you next year!


Fig. 9. On 24.04.2026 (WCAD), signatures of the attendees were collected on our "measurement instrument" (left - photo: A. Kołkowska); later on, some demonstrations of the sound of corrosion by A. Olesiński could be heard in the lecture hall (right - photo: A. Maciej)
Article author: Maciej Sowa
Original text: 23.04.2026
last update: 27.04.2026External references
- Rezić, I., Ćurković, L., & Ujević, M. (2009). Metal ion release from electric guitar strings in artificial sweat. Corrosion Science, 51(9), 1985–1989.
- Rezić, I., Ćurković, L., & Ujević, M. (2010). Study of microstructure and corrosion kinetic of steel guitar strings in artificial sweat solution. Materials and Corrosion, 61(6), 524–529.
- Rolich, T., Rezić, I., & Ćurković, L. (2010). Estimation of steel guitar strings corrosion by artificial neural network. Corrosion Science, 52(3), 996–1002.
- Rezić, I., Ujević, M., & Ćurković, L. (2014). Investigation of metal ion release from violin, viola, and cello strings after dissolution in corrosive solution. Materials and Corrosion, 65(9), 922–928.
- Borruto, A., Narducci, G., & Buccitti, M. (2013). Failure analysis of piano strings. Engineering Failure Analysis, 35, 164–177.
- López, J. L., Bonastre, J., Segura, J. G., Gadea, J. M., Juliá, E., & Cases, F. (2015). Correlations between acoustic and electrochemical measurements for metallic corrosion on steel strings used in guitars. Engineering Failure Analysis, 57, 270–281.
- Bonastre, J., López, J. L., Segura, J. G., Gadea, J. M., Juliá, E., & Cases, F. (2019). Cathodic protection of steel guitar strings against the corrosive effect of human sweat. Engineering Failure Analysis, 97, 645–652.
- Segura Alcaraz, J., Bonastre Cano, J. A., Juliá Sanchis, E., Gadea Borrell, J. M., & Cases Iborra, F. J. (2024). Evaluation of the acoustic performance of a novelty cathodic protection system against guitar steel strings corrosion. Heliyon, 10(9), e30811.